Yongbao Zhuang and Wenyang Shi: Scientific thinking with meaningful real-world impact
Doctoral researchers Yongbao Zhuang and Wenyang Shi are interns at the Medaffcon office in Stockholm.
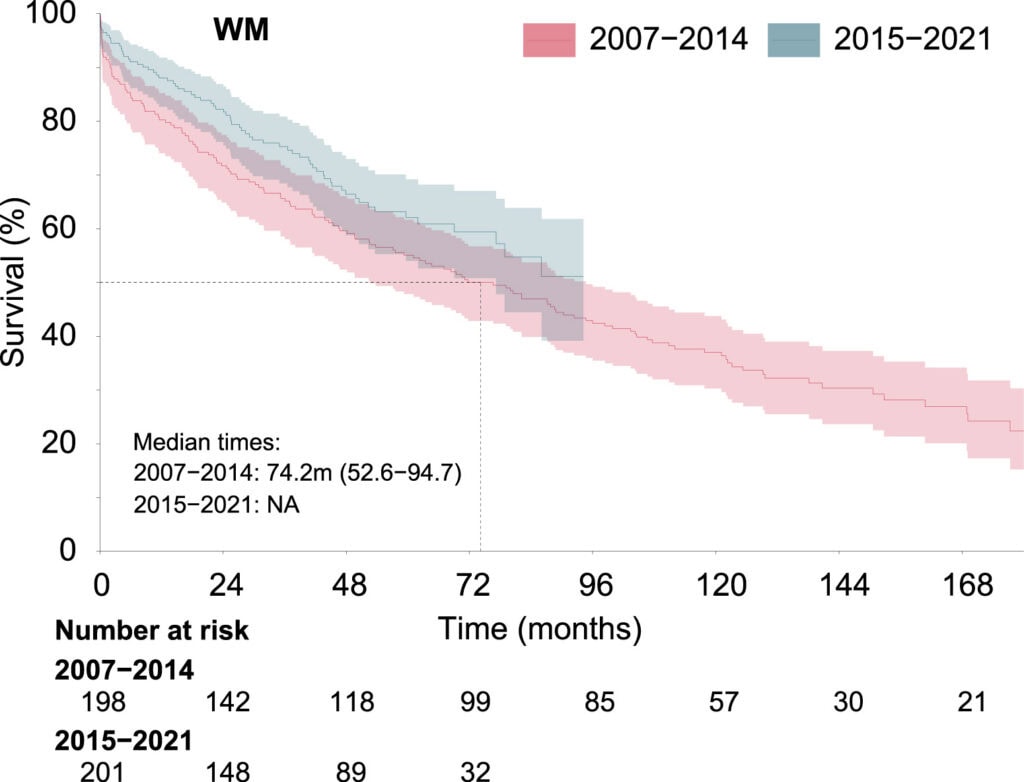
A national retrospective real-world evidence (RWE) study has generated significant new insights into the epidemiology, treatment, and prognosis of the rare blood cancer Waldenström’s macroglobulinemia (WM) in Finland between 2007 and 2021. The study, published in Clinical Lymphoma, Myeloma & Leukemia, is the first of its kind conducted in Finland.
The importance of the study lies not only in its findings but also in its clear demonstration of the value of registry data, particularly in rare diseases. Because patient populations are small and geographically dispersed worldwide, conducting clinical trials is often challenging.
According to Essi Havula, Scientific Advisor at Medaffcon, the findings align well with international publications. The study did not focus on detailed treatment choices. Instead, it examined incidence, patient characteristics, overall survival, causes of death, healthcare costs, and the proportion of patients receiving treatment.
“In diseases like this, registry research is invaluable when the goal is to understand patient characteristics and clinical outcomes. Registry studies are at the core of Medaffcon’s expertise, and this research demonstrates how much can be learned about a rare cancer using existing healthcare registries. Registry research can provide highly valuable information to support decision-making and the development of care,” Havula states.
Hematologist Pekka Anttila, the corresponding author in the study, also emphasizes the importance of registry data. In his view, registry data is often the only way to monitor and confirm that treatments perform as effectively in routine clinical practice as they do in clinical trials.
Registry information forms the foundation for evaluating the quality of care and planning future healthcare strategies.
“In more common diseases, registries enable comparisons between university hospital districts, for example. They allow us to assess whether treatments are delivered equitably across the country and whether outcomes are internationally comparable,” Anttila says.
He notes that particularly for new medicines, high-quality registry data can reveal both achieved benefits and previously unidentified adverse effects. This information is derived specifically from RWE studies—not solely from strictly controlled clinical drug trials.
Anttila emphasizes that high-quality data requires careful documentation. The quality of registry research depends directly on how accurately diagnoses and clinical information are recorded. The value of accurate recording should be pinpointed already in medical education.
“Entering diagnoses into healthcare systems is not always straightforward. In diseases such as Waldenström’s macroglobulinemia and multiple myeloma, a pathological sample alone is not sufficient—clinical assessment and diagnosis by a physician are required,” Anttila explains.
According to Anttila, the next study could focus on the new drug that has recently entered the market.
“Waldenström’s macroglobulinemia is extremely rare worldwide, so progress is inevitably slow. I would wait two to three years to see how the use of new medicines develops. A new drug has recently entered the market to compete with the existing one and comparing them could be interesting. The Finnish Social Insurance Institution (Kela) registries could provide ongoing data on medication use,” he notes.

Doctoral researchers Yongbao Zhuang and Wenyang Shi are interns at the Medaffcon office in Stockholm.

While the majority of Medaffcon’s revenue comes from the pharmaceutical industry, the company’s activities are not limited only to commercial projects. At any given time, several academic projects may be ongoing.

Digital solutions challenge established perceptions of what constitutes acceptable contraceptive methods and which health benefits are considered measurable.

Scientific Advisor
PhD
Essi joined Medaffcon in September 2023. Essi holds a PhD in biosciences and in her thesis she studied the genetics of sugar metabolism. After completing her pre- and postgraduate studies at the University of Helsinki, she transferred internationally to continue to study gene-diet interactions at the University of Sydney. Spanning over 15 years, Essi’s scientific career includes research in numerous fields including breast cancer and mitochondrial diseases.
Essi’s strengths include broad scientific and international background with special interests in personalized medicine, metabolic diseases and science popularization. At Medaffcon, Essi is particularly motivated by the utilization and communication of RWE studies to support better decision-making in our patient care and more broadly for our healthcare sector.
“Data driven decision making requires us as experts to process and communicate the vast amounts of data into a form that is accessible to all healthcare authorities. I’m looking forward to seeing how RWE studies can guide the development of personalized therapy solutions, that are not only economically the most efficient, but benefit the patients in the best possible manner.”