Target Trial Emulation brings the logic of randomized trials into real-world data
Target Trial Emulation does not replace randomized controlled trials, but it applies their logic and rigor to real-world data analysis.
While most applied transportability and generalizability research is conducted in the United States, Nordic real-world data offers pharmaceutical companies a largely untapped opportunity to extend evidence and support decision-making across the region.
In medical research, transportability and generalizability refer to the ability to apply evidence from a single study to different patient populations, clinical settings, or geographical regions for example to support HTA and reimbursement decision-making.
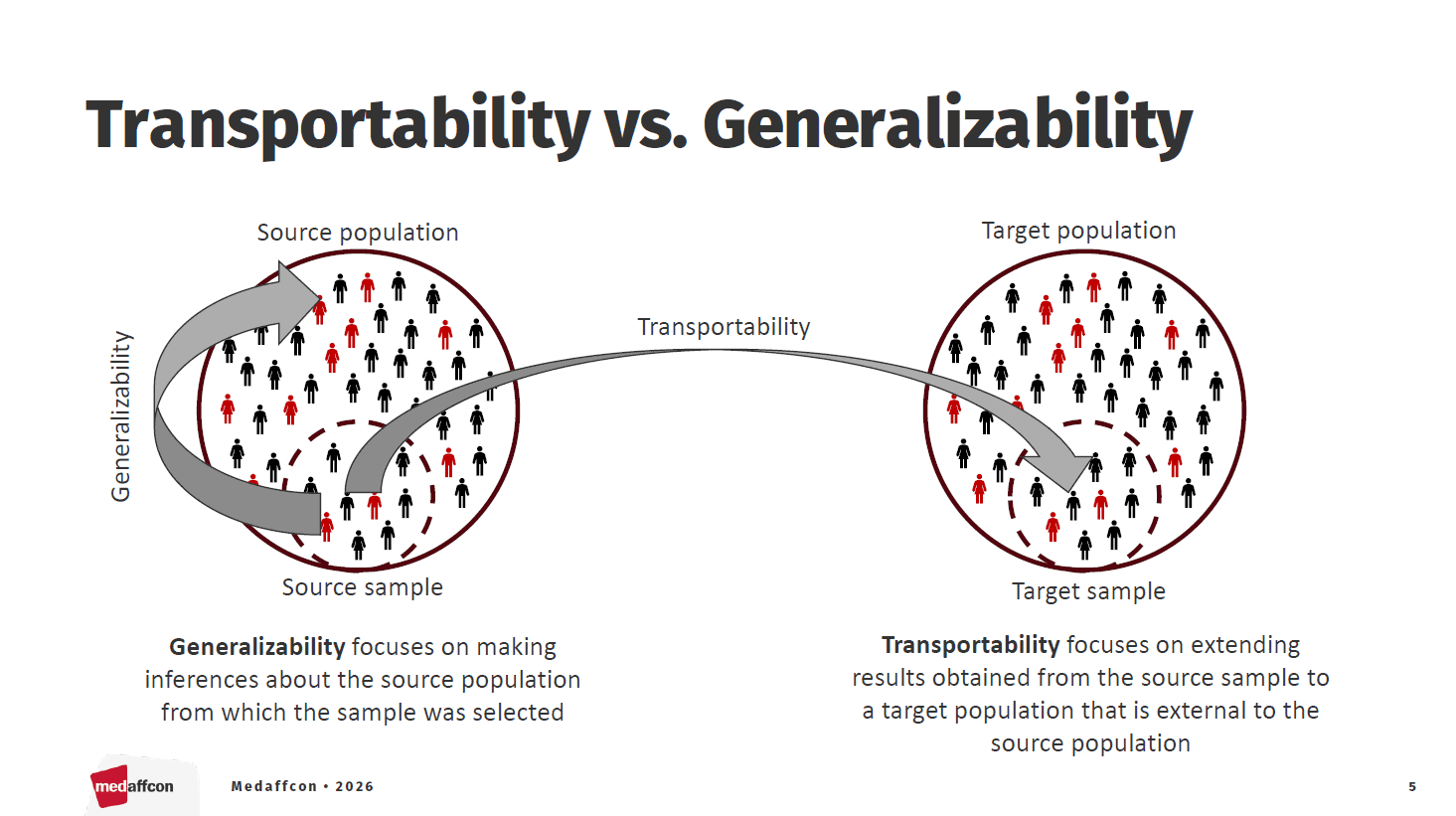
One particularly promising application is the use of external control arms to complement clinical trials and assess their relevance in local healthcare environments. Medaffcon’s Data Analysis Lead, Iiro Toppila, discussed the topic at the 6th Nordic RWE and AI Conference in Helsinki at the end of January.
“External control arms could be used more widely. They are especially valuable when there is a need to compare clinical trial outcomes with local standards of care and to ensure that the evidence supports decision-making in a specific healthcare environment,” Toppila says.
According to a systematic review published in Annals of Epidemiology, the number of studies applying transportability and generalizability methods increased fourfold between 2020 and 2024. Most of the research originates from the United States, with only a limited number of publications using European or the Nordic data.
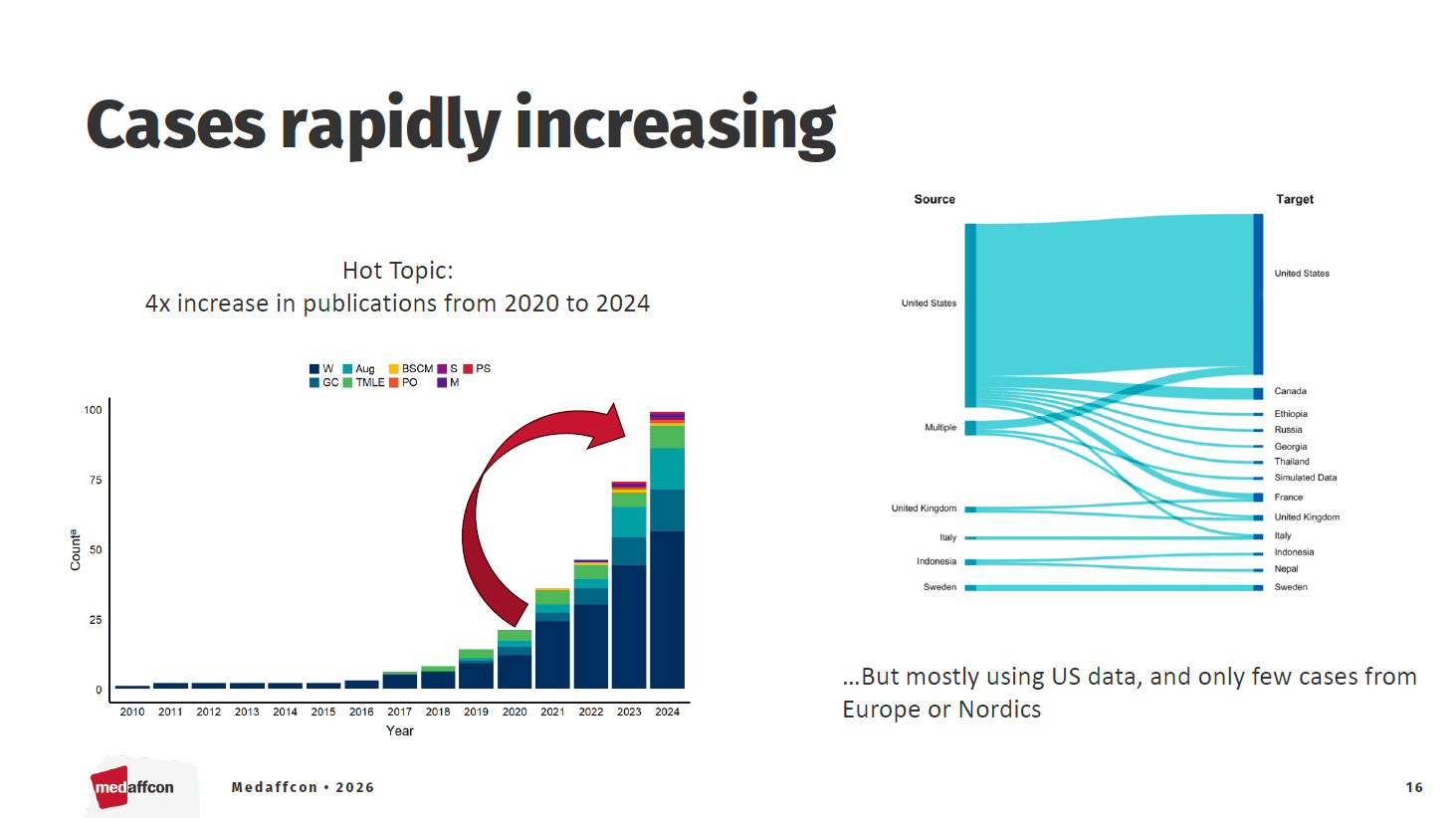
According to Toppila, this might be partly due to fewer regulatory barriers to the secondary use of health data in the US. However, tighter data governance in the Nordics does not prevent the application of these approaches.
“By applying methods that allow data to be analysed locally without cross-environment transfer, transportability and generalizability approaches can be implemented in a compliant way. A competent local partner plays a key role in identifying suitable methodologies,” Toppila explains.
Studies published to date provide strong use cases and demonstrate the feasibility of these approaches. Toppila notes that high-quality Nordic real-world data, particularly from Finland, offers an excellent basis for generating evidence that can be applied across Nordic and European healthcare settings and used in discussions with HTA bodies and pricing and reimbursement authorities. And vice versa, deep Finnish data would enable transporting external evidence into local Finnish setting. Medaffcon is currently conducting several studies with different transportability-focused approaches.
According to Toppila, the growing use of transportability and generalizability methods also highlights the need to carefully assess the validity and reliability of the resulting evidence.
To use these approaches effectively, pharmaceutical companies must plan evidence generation at a Nordic or multi-country level and initiate projects early, as study design and data preparation are time-consuming. Where existing data sources, such as Medaffcon’s CORE dataset, can be leveraged, timelines can be significantly accelerated, Toppila concludes.
Systematic review of applied transportability and generalizability analyses: A landscape analysis. Vuong Q, Metcalfe RK, Ling A, Ackerman B, Inoue K, Park JJH. Annals of Epidemiology (2025).
Matching techniques go beyond virtual control arms, reminds senior data scientist at Medaffcon
Virtual control groups in pharmaceutical development – What are they and can they be trusted?
Target Trial Emulation brings the logic of randomized trials into real-world data

Target Trial Emulation does not replace randomized controlled trials, but it applies their logic and rigor to real-world data analysis.

The data team keeps Medaffcon's research projects on track and ensures that the research findings are scientifically sound. At the heart of the team’s work is the processing and analysis of patient data, particularly in RWE studies.

The algorithm was originally developed to extract smoking status from patient texts with purpose to analyze the effects of smoking on postoperative complications. Today, it is also being utilized in lung cancer research.

Data Analysis Lead
MSc (Tech.)
+358 44 314 1597
iiro.toppila@medaffcon.com
Iiro joined Medaffcon in March 2017 as a Biostatistician. For the preceding four years, he has worked as a research assistant in an academic study group, analyzing clinical and genetic patient data. Iiro holds a Master of Science degree in Technology in Bioinformation Technology.
Iiro’s strengths include his strong expertise in statistics and data-analysis, hands-on experience in working with sensitive patient data, and strong interdisciplinary communication skills with experts from various fields. In the field, he is particularly interested in the large data amounts made available with the revolution of technology and how the information received such data can potentially be utilized to draw concrete conclusions, both in order to understand the nature of diseases and to advance the goals of the pharmaceutical industry and patient treatment.
“Machine learning and AI-based solutions will have a major impact on the healthcare sector now and in the future. However, effectively utilizing the already collected and available health-data will have a higher importance in order to improve health-care”.